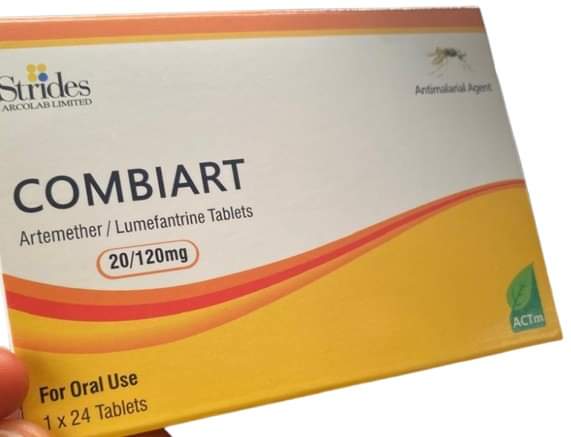
THE National Agency for Food and Drug Administration and Control (NAFDAC) has informed the public of the circulation of counterfeit Combiart dispersible tablets 20/120mg in the country.
The product, manufactured by Strides Arcolab Limited, which is based in India, was discovered in the Federal Capital Territory (FCT) and Lllal, Rivers State during surveillance activities by NAFDAC.
According to NAFDAC, the Laboratory report of the analysis carried out on the product revealed that it contained Zero APIs. The product was also observed to have two different date markings. The NAFDAC database for registered products has confirmed that the product license has expired and the NAFDAC Registration Number is wrong and not for the product.
Product details –
Brand Name: Combiart Dispersible Tablet 20/120mg
Generic Name: Artemether + Lumefantrine 20/120mg Dispersible Tablet
Batch No: 7225119
NAFDAC Reg No: A11-0299
Manufacturing Date: June 2023 and Feb 2023
Expiry Date: May 2026 and June 2026
Manufacturer’s Name and Address: Strides Arcolab Limited, 36/7, Suragajakkanahalli, Indlavadi Cross, Anekal Taluk, Bangalore- 562 106, India.
Healthcare professionals and consumers are advised to report any suspicion of the sale of substandard and falsified medicines or medical devices to the nearest NAFDAC office, call 0800-162-3322 or send an email to sf.alert@nafdac.gov.ng
Eighteen-Eleven Media